In a notice on Jan 9, the MFDS listed the identities of the social media influencers, the types of products involved, and the types of offence.
The MFDS has been monitoring influencers with over 100,000 followers on social media platforms and have been advertising weight-loss supplements on their accounts for five months since last August.
It found 153 problematic adverts concerning 33 weight-loss products, including ‘detox juice’ and teas.
False and exaggerated ads were the most common problem with influencer-advertising, with 65 cases found.
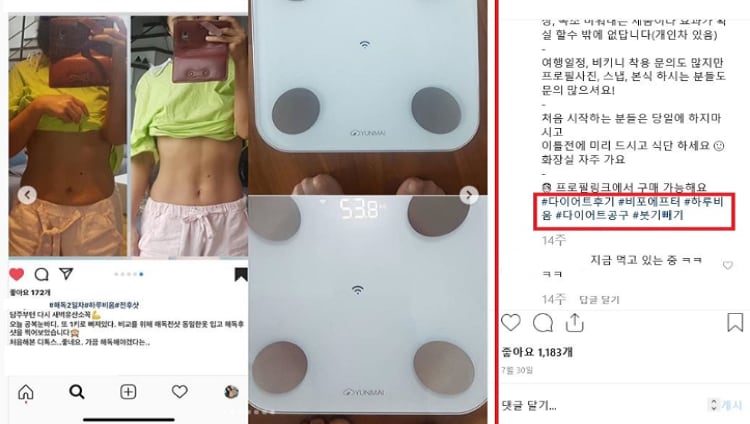
Experience-based ads which claim product efficacy using before-and-after photos were the next biggest problem with 34 cases found. The MFDS pointed out that these photos were mostly photoshopped.
Another five even claimed that the supplements could treat and/or prevent diseases.
The ministry also raised concerns about the lack of clinical evidence behind these products.
The MFDS said they would continue to look out for fraudulent advertising.
It added that even if no product selling was involved, the influencers could still be subjected to criminal punishment if they posted videos or photos which made dubious claims.
"The crackdown on social media platforms took long because it was hard to trace the users' personal information.
“However, the food safety ministry will keep monitoring those kinds of false advertisements on social media to protect customers' health and safety," Korea Times quoted Shim Jin-bong, the deputy director of the MFDS' cyber investigation bureau.
With the rise of social media, it is increasingly common for firms, including health foods brands to engage influencers in promoting their products.
In South Korea, over KRW$2tr (US$1.86bn) was spent for advertising on mobile platforms in 2017, surpassing that of cable TV (US$1.71bn) for the first time, according to a report from The Korea Herald.
This is especially so in China where companies engage key opinion leaders (KOLs) to talk about their products on platforms such as WeChat, Tik Tok, and the Little Red Book.
Stringent control
The South Korean authorities have scheduled a series of actions to beef up the quality control of dietary supplements.
For example, manufacturing processes are required to follow Good Manufacturing Practices (GMP) from December.
From May, the authorities will also start to investigate the causal effects of dietary supplements producing abnormal side effects.