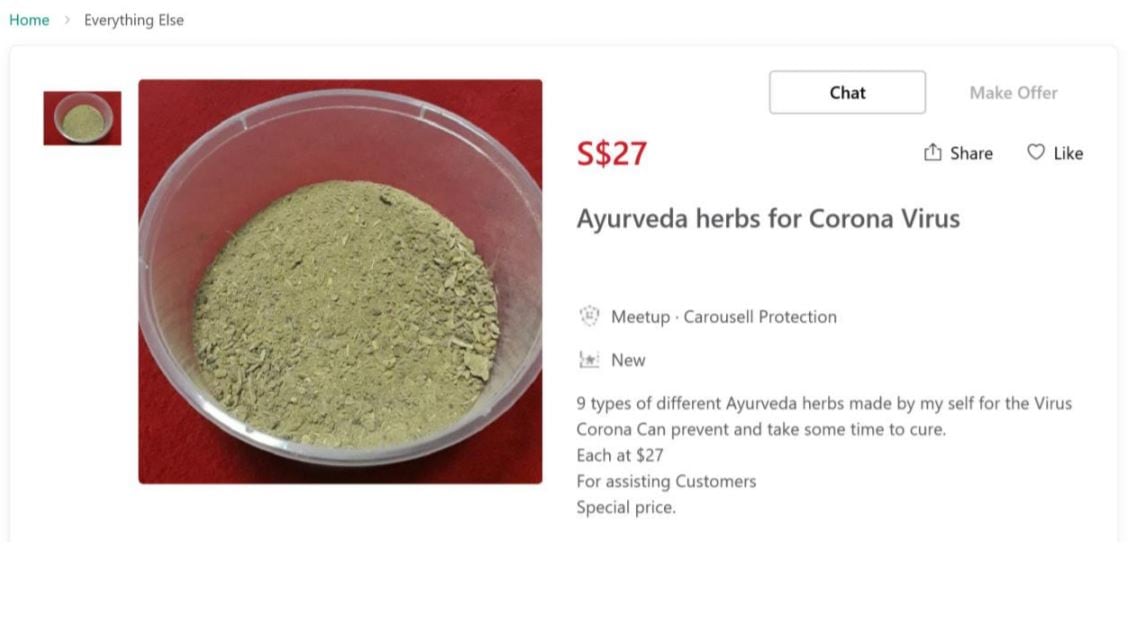
These fraudulent products included health supplements, herbs, traditional medicines and “clip-on” products.
According to HSA, these products contained false and misleading claims, such as “strengthen the immune system against the coronavirus” or “prevent and cure coronavirus”. There is currently no evidence that such products can prevent or treat COVID-19.
In an official document, HSA referred some of the products that had been associated with false claims.
However, these claims were made by the e-commerce sellers, and not the brands themselves.
More than half
The clamp down on fraudulent products was part of HSA’s involvement in an Internet-based enforcement action (Operation Pangea) coordinated by Interpol.
A total of more than 2,500 listing of adulterated products between March 3 to 10 were removed.
Product listings with false claims related to COVID-19 made up about half, while adulterated lifestyle products such as weight loss pills, sexual enhancement medicines and cosmetic products comprised more than 32% of the listings taken down.
In addition to health products, HSA also detected false rapid test kits which claimed to diagnose COVID-19 in just 10 minutes. They explained that “Testing for COVID-19 can only be done by clinical laboratories or medical professionals in clinics and hospitals to ensure an accurate test result and diagnosis.”
No science, don’t sell
The HSA reiterated that “There is currently no evidence that any health supplement, Chinese proprietary medicine, traditional medicine, herbs or “clip-on” product can boost the immune system specifically to help prevent, protect against or treat COVID-19.”
Adding that some products may claim to be developed based on scientific studies or evidence, but such claims often lack a robust scientific basis and cannot be verified.
They advised consumers who are planning to purchase health products online, to do so through websites with an established retail presence in Singapore.
Singapore has some of the world’s strictest punishments, with those engaging in the sale or supply of adulterated health products or carry misleading claims, liable to prosecution and if convicted, may be imprisoned for up to three years and/or fined up to SGD100,000 (USD69,000).
The U.S. Food and Drug Administration (FDA) and the Federal Trade Commission (FTC) have also recently issued warning letters to seven companies for selling fraudulent COVID-19 products. The products cited in these warning letters included teas, essential oils, tinctures and colloidal silver.
In an official statement, FTC’s chairman, Joe Simons said: “There already is a high level of anxiety over the potential spread of coronavirus. What we don’t need in this situation are companies preying on consumers by promoting products with fraudulent prevention and treatment claims.”
Globally, there have been more than 300,700 confirmed cases of COVID-19 and more than 13,900 deaths at the time of reporting (March 23).