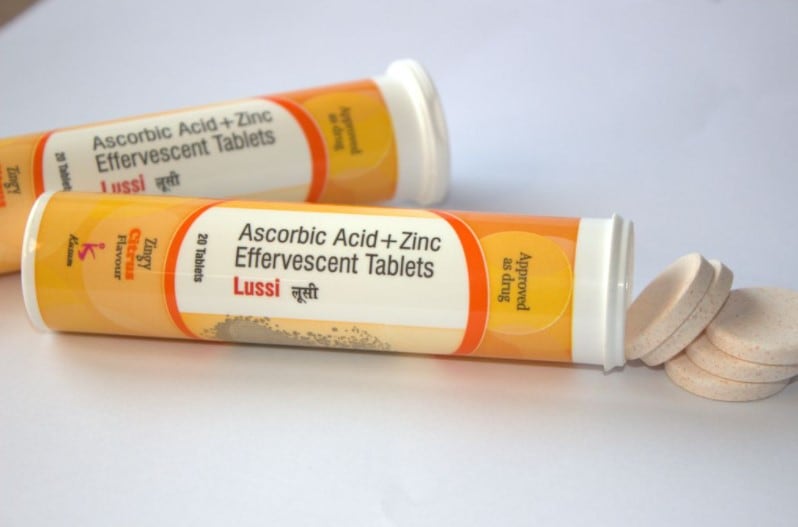
Launch under the Lussi brand, the supplement comes in effervescent tablets which combines 1,000mg of ascorbic acid and 10mg of zinc.
Kusum Healthcare researches and manufactures more than 500 medicines and supplements exported across 30 countries. Lussi is the latest product launched after the firm saw the need for a strong immunity supplement amid the pandemic.
According to Nimesh Nayak, assistant general manager of domestic marketing at Kusum Healthcare, although vitamin C supplements are readily available in India, yet most of the vitamin C was derived from plant extracts.
While plant extracts offer the advantage of being natural, its bioavailability is often low, he claimed, adding that 1,000mg of vitamin C derived from plants or herbs, may only deliver 40mg in the human body.
“Our R&D team found that vitamin C developed in synthesised form will give a larger dose of vitamin C to humans.”
Kusum Healthcare is currently applying a patent for its manufacturing process which promises a vitamin C bioavailability of more than 90%.
Synthetic vitamins are formulated in a lab setting, while natural vitamins are derived from plants, animals or other natural sources and then manufactured in the lab.
Synthetic vitamin C is not new, with vast amounts of ascorbic acid synthesised yearly by the pharmaceutical industry.
However in India, Lussi is the first local brand to produce an immunity supplement entirely synthesised in the lab without any plant extracts.
According to Nayak, its supplement cannot be considered a nutraceutical since nutraceuticals tend to contain plant extracts.
“Vitamin C and zinc supplements are plenty in the market, so we tried to carve a niche for ourselves by creating a differentiation in terms of our ingredients, and ultimately making sure the end consumer receives the maximum benefit of our product.”
As a supplement, it is available at offline and online pharmacies direct for consumers.
Doctor channel
Lussi is also approved by the Drugs Controller General of India (DCGI) as a drug, which allows the firm to promote the product to doctors and other healthcare professionals.
The application process to be approved as a drug did not require any clinical studies, instead it is based on available data on vitamin C and zinc, for which the concept is already well-established.
Although, the process requires Lussi to submit a post-marketing surveillance report to DCGI, in which the firm is currently working on a database of Indian patients.
Since the launch of Lussi about two months ago, it has sold almost 20,000 units to more than 1,000 doctors, and the pharmacy retail channel such as Tata 1mg.
Lussi is also working on building its own e-commerce store.
“Because it is the first-of-its-kind product in India, we initially launched through doctors who can educate patients,” Nayak said.
“As we gain more acceptance overtime, we plan to enter the dermatology field because vitamin C is also known to help with skin-related issues.”
According to Kusum’s head of business optimisation Upasana Gupta, Lussi will export the supplement to Ukraine and the Philippines early next year.