Australian regulator the Therapeutic Goods Administration (TGA) has said 7-Days Herbal Slim-Extra capsules pose a serious health risk and should not be taken.
Its tests revealed that the capsules contain the undeclared substance sibutramine — the active ingredient in Reductil. It was withdrawn in October 2010 after a study showed it was involved in an increased risk of major cardiac events.
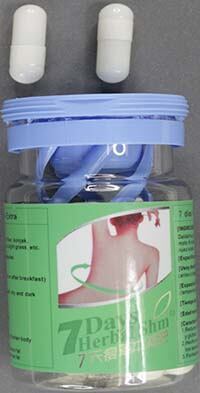
The TGA said: "The supply of 7-Days Herbal Slim-Extra capsules containing undisclosed sibutramine is illegal. 7-Days Herbal Slim-Extra capsules have not been assessed by the TGA for quality, safety or efficacy as required under Australian legislation, and the place of manufacture is not approved by the TGA."
Online trading
The TGA's investigations have shown that a number of people in Australia may have bought the product online. Consumers have been urged to stop taking the supplement, and bring any remaining capsules to a pharmacy for disposal.
The product appears to originate from China, with the firm's website claiming: "7 days herbal slim is made from extracts from Seville orange flower (daidaihua) and lucid ganoderm (lingzhi) in Yunnan botanical kingdom."
The TGA is now working with the Australian Border Force (ABF) to help stop future shipments from entering Australia.
If these capsules are found at the border by the ABF, they will be seized and destroyed.

