This is a breakthrough in the current market, as oral supplements that claim to increase the body’s NAD+ levels mostly work through NAD+ precursors, such as nicotinamide mononucleotide (NMN) or nicotinamide riboside (NR), instead of delivering NAD+ directly into the body.
These precursors are a form of vitamin B3.
Entity Health launched the new product in Singapore last month through its webstore and will be launching it in China via its Tmall flagship store.
Known as “SL-NAD+”, the product is an addition to the brand’s existing NAD+ precursor product range, which consist of RestoriX, which contains 250mg of nicotinamide per tablet and MetaboliX Plus, which contains 250mg of nicotinamide per capsule.
As a NAD+ precursor, nicotinamide, as well as NMN and NR, will need to undergo biological processes in the body so that they will be converted into NAD+ for use in the body.
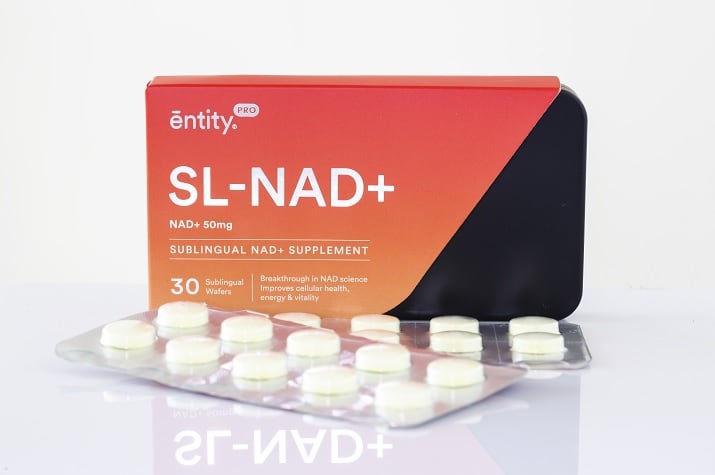
In contrast, the new supplement, which contains 50mg of NAD+ in the form of sublingual wafer, is said to deliver NAD+ directly into the body, hence skipping the conversion process. As a sublingual wafer, the product is meant to be placed under the tongue during consumption.
The product was developed using a patented wafer technology known as WaferiX – which is owned by Singapore-based iX Biopharma, the company which also owns Entity Health.
As NAD+ is a highly reactive co-enzyme, the product needs to be refrigerated and transported by cold chain.
Why made this?
Speaking to NutraIngredients-Asia, Dr Janakan Krishnarajah, chief medical officer at Entity Health, said that the company's nicotinamide supplements have been well-received, especially in China, and that the Chinese consumers were seeking for products that contained NAD+ itself.
The two existing nicotinamide supplements, RestoriX and MetaboliX Plus, were among its top three bestsellers in China, alongside its glutathione supplement LumeniX, he said.
“What we have seen is that the consumers are getting more educated and more sophisticated. They are looking for the next 'in' thing and new premium products that are available out there.
“We have also noticed that people are looking at NAD+ itself and we have seen these queries coming out of China, and so, we have started to focus on whether we could develop a product that contains the actual NAD+ molecule in its purest form.
“We have launched this product to give consumers another choice and this will be a premium choice because of the nature of the NAD+ moleucule and the delivery technology used.”
Each box of “SL-NAD+” contains 30 wafers and will retail at SGD$188 (US$139), with discounted price given during the launch.
On the other hand, RestoriX is retailing at SGD$39 (US$28.95) for a bottle of 60 tablets, and MetaboliX Plus is retailing at SGD$49 (US$36.38) for a bottle of 60 capsules.
Dr Krishnarajah said that consumers generally understood the function of NAD+.
“The consumers understand that as NAD+ level decreases, they will be more susceptible to diseases and that NAD+ could slow down the process of DNA damage and provide inflammation control.”
The “SL-NAD+” product claims to “improves cellular health, energy, and vitality.”
Prior to the launch of this product, he said that the other method to deliver NAD+ directly into the body was through intravenous injection.
Behind the scenes
The process of developing the NAD+ supplement took almost two years, with the team overcoming challenges around finding the right raw material supplier, ensuring product stability, and maintaining the product’s freshness.
Dr Krishnarajah declined to reveal the supplier or the manufacturing process of the NAD+ ingredient. Nonetheless, he explained that NAD+ was a natural molecule produced in the body and that when made into an oral supplement, it could generally be created via synthetic processes using amino acids or vitamin B3 as the starting raw material.
At the moment, “SL-NAD+” has a shelf-life of 12 months and the company is now working on expanding it to two years.
Using the WaferiX wafer technology – the proprietary delivery technology stabilises NAD+ and the excipients in the sublingual wafer form.
“We have been able to do that because we have a unique technology, a patented delivery technology call WaferiX, which makes freeze-dried sublingual wafer that is to be placed under the tongue,” he said.
As NAD+ is a highly reactive co-enzyme, it would be affected when exposed to heat, which is why refrigeration is required.
The product is manufactured in a facility certified by the Therapeutic Goods Administration (TGA) in Australia.
More fast acting?
As the product delivers NAD+ directly, it is said to be more fast-acting than the other NAD+ products, according to Dr Krishnarajah.
“We are delivering pure NAD+ directly into the bloodstream as the sublingual wafers will disintegrate under the tongue, and yes [it will be more fast-acting], because NAD+ is going directly into the bloodstream,” he explained.
In contrast, NAD+ precursors, when consumed as a oral supplement, will be broken down in the small intestine to nicotinamide, ribose, phosphate, and adenosine before being absorbed into the bloodstream.
These molecules are then reassembled by the cells, forming NAD+.
“This process, however, is mainly dependent on the individual’s ability to reassemble the molecules into NAD+; hence improvement to the body’s NAD+ levels may vary for each person.
“Some of these NAD precursors are also partially broken down and lost when they enter the digestive GI tract. Furthermore, not all cells are capable of reassembling precursors into NAD+,” the firm explained.
Clinical trial
The company is in the process of designing a human clinical trial to assess the effects of NAD+ oral supplementation in improving metabolic health.
The trial is expected to commence in Singapore next year.