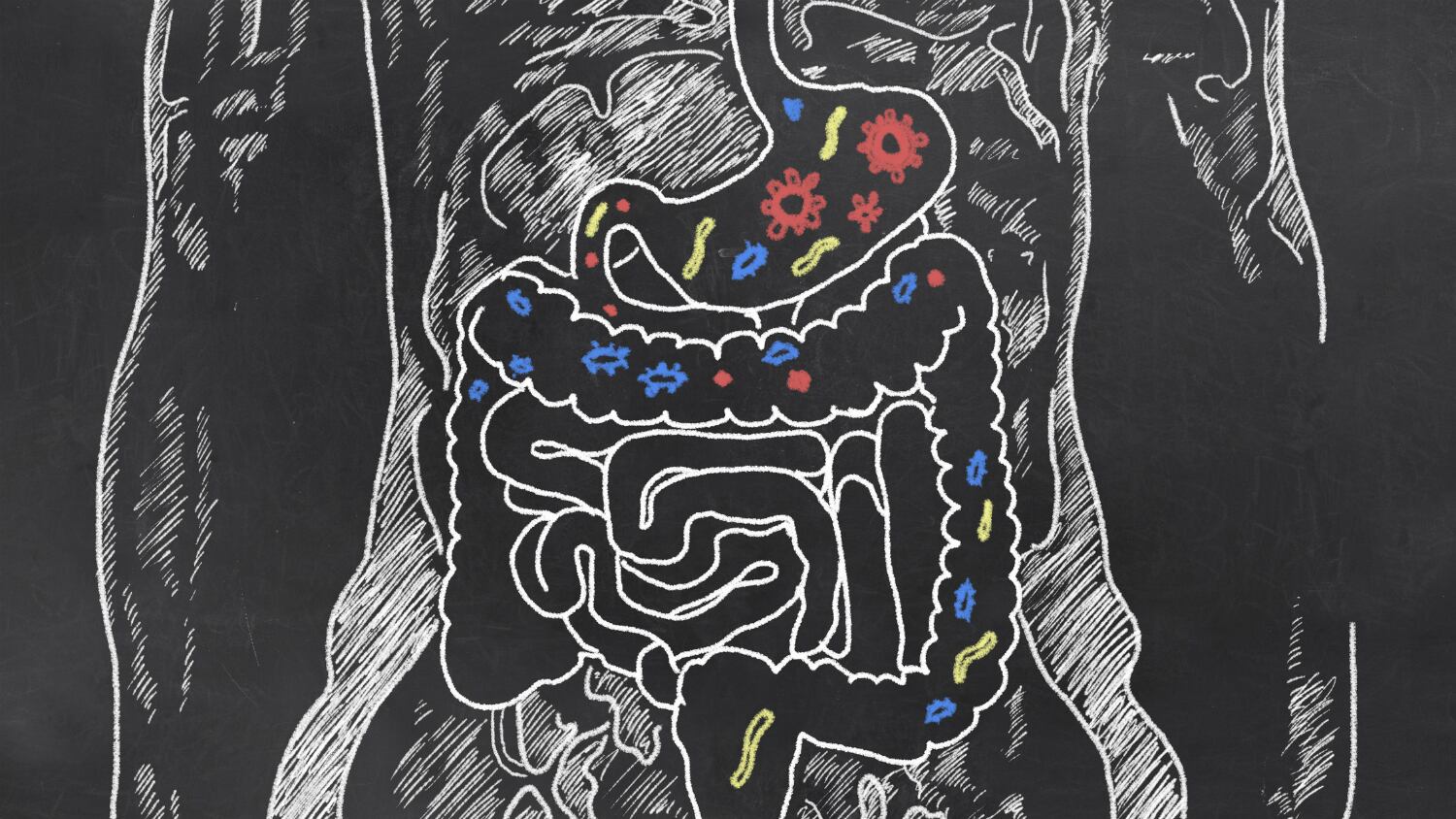
Scientists at New Zealand's Plant & Food Research institute, together with local and international research partners, have discovered a gut bacterium that specialises in breaking down pectin, a carbohydrate found in plants that is typically difficult to digest.
Degradation specialist
Called Monoglobus pectinilyticus, the bacterium is said to be the first to specialise in pectin degradation and utilisation.
Pectin, a primary source of dietary fibre in human diets, also acts as a plant's natural barrier against bacterial attacks — it comprises 40% of the plant cell wall in fruits and vegetables like kiwifruit and tomato.
Given its structural complexity and low energy content, most bacteria are unable to break down its barrier in order to access and digest the plant material they need to survive.
Prior to this study (published in The ISME Journal), the process by which pectin is broken down was not well understood — only a few species of pectin-degrading bacteria exist, and none are as specialised as M. pectinilyticus.
Plant & Food Research's Dr Caroline Kim, who leads the project, said M. pectinilyticus' high degree of specialisation suggested that the human body’s regular pectin intake may have placed 'evolutionary pressure' on the gut microbiome to accommodate specialist bacteria dedicated to pectin degradation.
Kim and her team analysed the dietary intakes and faecal samples of 44 healthy individuals in New Zealand over the course of 10 weeks, and found that the abundance of M. pectinilyticus was positively associated with the subjects' pectin consumption.
She told NutraIngredients-Asia that pectin is not a palatable substrate for most bacteria, as it is difficult to break down but produces little energy.
"M. pectinilyticus is special in that it invests a remarkable 45% of its carbohydrate-degrading enzymes into degrading pectin, making it a 'specialist'. In contrast, (other pectin-degrading bacteria, such as) Bacteroides spp. are 'generalists' as they degrade many different types of carbohydrates, but invest a relatively small portion of their enzyme pools for degrading pectin.
"As M. pectinilyticus only degrades pectin, we may be able to use it as an 'interference-free' model organism that provides an uninterrupted molecular view of the colonic pectin degradation. It will be highly useful when studying pectin degradation in isolation, something that was hard to do using generalist bacteria."
She added that the composition of M. pectinilyticus' pectin-degrading enzymes resembled that of plant pathogenic bacteria more closely than human gut bacteria, suggesting that it could aggressively attack pectin to help penetrate the plant cell wall.
Furthermore, M. pectinilyticus cells bind tightly to plant cell wall pectin, a trait previously unobserved in human gut bacteria.
Elucidating mechanisms
Kim said: "How M. pectinilyticus binds to and breaks down pectin is our next big research question. So far, we know it does not use any of the most-studied paradigms for carbohydrate degradation by human gut bacteria — it is phylogenetically separate from any human gut bacteria, and its closest relatives — though still very distantly related — are cellulose-degrading environmental bacteria."
Curiously, however, M. pectinilyticus has evolved to focus on pectin rather than cellulose, leading Kim to predict that humans may have acquired M. pectinilyticus from the outside environment long ago, and that the strain possibly developed pectin-degrading characteristics to cope higher pectin intake in human diets.
She also said there would be further research, as there was still much to discover about how M. pectinilyticus broke down pectin, if there were more of these organisms colonising the human gut, and if they used similar a carbohydrate degradation mechanism to break down pectin and / or a broader range of substrates.
"The primary outcome (of such a study) would be to understand the underlying molecular biology that enables M. pectinilyticus to be an efficient pectin degrader.
"We also want to see if M. pectinilyticus is a 'keystone' bacterial species (i.e., less pectin degradation occurs in the absence of this organism in the human gut) or if humans harbour alternative and redundant pectin-degrading organisms similar to M. pectinilyticus.
"It would also be interesting to compare the presence or absence of M. pectinilyticus in different human populations consuming high- or low-fibre diets."
Cultivation and commercialisation
In light of emerging research around new bacterial species in the human gut microbiome, Kim said the microbial diversity and complexity of the human gut had been shown be much greater than originally anticipated.
As such, it would take scientists a long time to discern the impact of these microbes on human health at species / strain levels.
In terms of the commercial implications of this line of research, Kim said: "We have considered the commercial potential of M. pectinilyticus, but it's a very slow-growing microorganism, so we don't think it would be practical to use it in probiotics or in any large-scale production.
"Commercially, it might be possible to genetically engineer its enzymes, because companies like Megazyme are always interested in buying new families of enzymes that can fulfil new functions."
Such commercialisation will not materialise anytime soon, however. Researchers in this field are largely focused on cataloguing any new bacterial species before they can elucidate their functions in the human gut.
Furthermore, bacteria are notoriously difficult to cultivate, and there is a dearth of information needed to effectively incorporate the microbiome into therapeutic applications or commercial ventures.
Kim said: "We've been trying to work in the reverse instead of trying to figure out how plant material can change the microbiome when consumed in human diets. We know pectin-degrading bacteria exists, but we don't really understand yet how it breaks pectin down.
"We want to see if we can use this information to screen vegetable and fruit cultivars that are more accessible to the gut microbiome and therefore, more easily digested by humans."