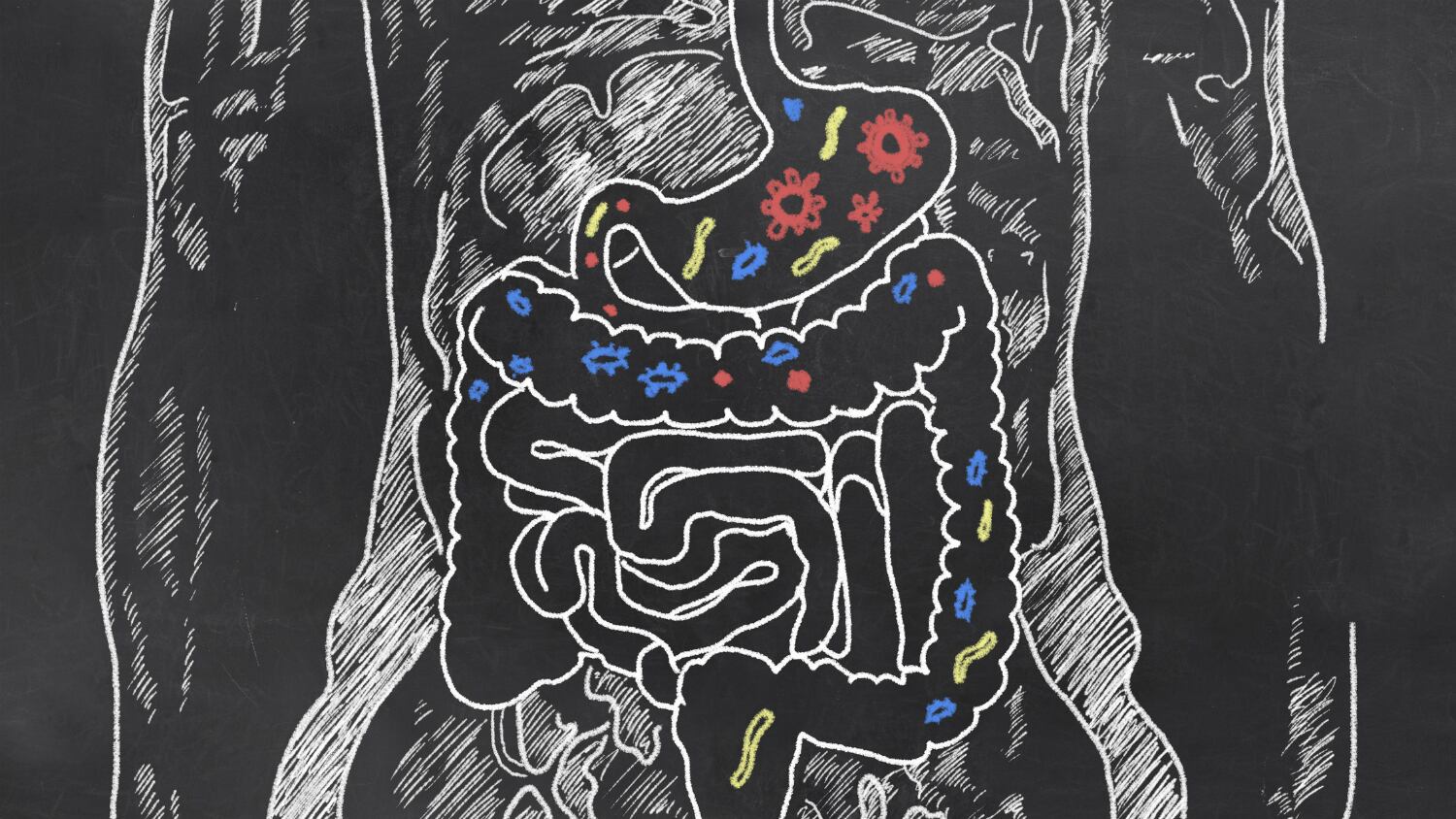
The study — conducted by Australia's Hudson Institute of Medical Research and Monash University, the UK's Wellcome Sanger Institute, and the European Molecular Biology Laboratory's European Bioinformatics Institute (EMBL-EBI) — entailed isolating the newly discovered species from the intestines of 20 healthy individuals from the UK and Canada.
The researchers studied the participants' faecal samples, from which they successfully grew and DNA-sequenced 737 individual bacterial strains. They then identified 237 separate bacterial species from these strains, among which were 173 species that had not been sequenced before. Of these 173,105 species had never been previously isolated.
The results of the study have been published in Nature Biotechnology.
Improvements in isolation
So far, the high level of difficulty in growing bacteria in a laboratory setting has translated to large gaps in the available information and knowledge about the different species of gut bacteria.
Standard methods employed include genome and DNA sequencing from mixed gut bacteria samples, but the lack of individually isolated bacteria and their reference genomes have restricted the level of information scientists can gather from them.
This new and improved collection of bacteria, however, is expected to vastly improve research into the human microbiome by facilitating quicker and more accurate detection of bacterial strains in the gut.
Speaking to NutraIngredients-Asia, lead author Dr Samuel Forster (from Hudson Institute, Monash University and Wellcome Sanger Institute) said: "We've discovered a lot more bacterial diversity than we had expected to find.
"The key thing we did was genome sequencing, and we managed to keep the bacteria alive, so we can conduct experiments to see what they might be doing in their interactions with people, as well as other bacteria we may find in the gut. This opens up a whole new type of research we can conduct on the microbiome.
"Previously, a lot of the work depended on sequencing and looking for associations. What we can do now is identify new bacteria faster by looking at the genome and determining which species or strains are closely related, or even identical."
He added that having this information would then allow scientists to conduct experiments to determine the biology of these bacterial strains, calling it a 'game-changer' for how microbiome research was conducted.
While he acknowledged that there were still thousands of species of bacteria that had yet to be isolated, he said it was encouraging that scientists had started to 'make a dent' in those numbers.
In an official release, co-author Dr Rob Finn (from the EMBL-EBI) called the database of reference genomes from pure gut bacteria isolates 'crucial', saying, "If they (researchers) want to test a hypothesis — for example, that a particular species is enriched in a certain disease — they can get the isolate itself from the collection and physically test it in the laboratory."
Dr Trevor Lawley, senior author from the Wellcome Sanger Institute, said: "By culturing the unculturable, we have created a resource that will make microbiome analysis faster, cheaper and more accurate. Ultimately, this will lead us towards developing new diagnostics and treatments for diseases such as gastrointestinal disorders, infections and immune conditions."
Forster added: "Growing bacteria is challenging as they have very strict nutrient requirements and are very sensitive to oxygen. But we have demonstrated in our previous work that it's possible, and this is the application of that work."
Probiotic potential
The results of the study not only indicate great potential for NPD and therapy for those with gastrointestinal and immune conditions, but also further discovery of more gut bacteria strains.
Forster said: "It's important to remember that all the work published in the Nature Biotechnology study was based on the microbiome of just 20 individuals, so we obviously haven't captured the intestinal diversity of people yet.
"At this stage, we're still trying to identify the more important bacterial strains, but this gives us a library of potential treatment. We just have to work out which strains are more likely to be useful."
He added there was also a 'huge amount of potential' in spaces like probiotics, and bacteria therapy.
However, in order for the commercial potential of useful bacterial strains to be properly realised, it would be necessary to further improve the culturing of these strains.
"We've generated a media that can replicate what you would expect within the human gut environment, but it would be ideal if we could find a way to develop a media that would help to improve growth rate of the species of choice."
He said there were two directions this research would take: one would be to continue building the bacterial collections so there would be more species to work with, which would in turn mean more potential products and therapeutic opportunities.
The other would be to apply this knowledge in a way that would make clear whether the absence or presence of a particular bacterial strain was associated with a certain condition. This way, the direction of commercialisation and further testing can be easily determined.
Forster further revealed that he and his fellow researchers intended to conduct similar studies within the Asia-Pacific region.
"We've already started the process in Asia-Pacific. In Australia, we've started collecting samples, and we are looking to expand what we have to a global collection over the next year or so."